Laboratory of gas chromatography
Head of Geochemical Laboratories Unit: Manfredi LONGO
E-mail: manfredi.longo@ingv.it
Referent of Gas Chemistry Laboratory: Walter D’ALESSANDRO
E-mail: walter.dalessandro@ingv.it
E-mail: laboratoriogascromatografia.pa@ingv.it
Laboratory of gas chromatography
Gas chromatography is an analytical technique which allows to separate, one by one, the different components of a mixture. It can be applied to gas samples, liquids and solids, provided that they can be transferred to the vapor state within certain temperature ranges. One of the typical applications of gas chromatography applied to Earth Sciences, consists in the analysis of gaseous and liquid phases in volcanic active areas, where the study of the volatile components provides valuable information about the physical and chemical conditions and on the dynamics of deep magmatic fluids.
The operation of a gas chromatograph can be simplified into the following steps.
The sample to be analyzed is subjected to the flow of a carrier gas (mobile phase), usually consisting of an inert gas, such as nitrogen, helium or argon, and introduced within a chromatographic column. Inside the column there are substances (stationary phase) able to separate the various components of the gaseous mixture. Along the column, the different constituents of the sample are slowed down to varying degrees and then separated.
At the end of the column, one or more detectors report the arrival of the different components to a signal processing system. The final product is a gas chromatogram where the various components of mixture appear as peaks in function of time.
The three main parts, of which a gas chromatographic system is made of, are shown schematically in Fig.1
1) A carrier gas cylinder fitted with pressure reducers and filters, these latter able to purify the inlet gas from any water vapor and various impurities;
2) A properly so-called gas chromatograph, with a further flow regulator, a system of sample introduction, one or more columns, one or more detectors;
3) An interface that connects the gas chromatograph to a software for the management, processing and storage of data.
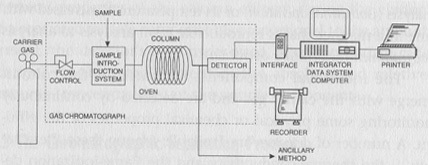
Fig.1
The gas chromatography laboratory of the INGV-Palermo is able to perform quantitative chemical analysis of free gases, gas enriched by removal of CO2 in ampoules of NaOH and gas dissolved in water.
The concentration of the samples is determined on the basis of the external standard method, by means of gas standards specially purchased, with concentrations of the various species varying between a few ppm and 100%. Appropriate calibration curves return the concentration values of the various analytes. A good evacuation of the input loop and of the whole introduction system is guaranteed by rotary pumps.
The gas introduction in gas chromatographs takes place through a properly designed system (Fig.2), that brings the sample to a multiport valve from which subsequently the sample will be introduced into the column. In this configuration, here is no need to use injectors. Obviously, when using He as carrier gas it is impossible to investigate the He concentration, and the same applies to the Ar.
The detectors used are:
A) Thermal conductivity detector (TCD), also called Hot Wire Detector (HWD). It is a detector sensitive to the concentration of the analyte, universal and non-destructive. Its operation depends on the different thermal conductivity of various gaseous species (Fig.3).
It consists of two strands, one crossed by pure carrier, the other by the gas outgoing by column (carrier plus sample). The filaments are part of a Wheatstone bridge (balanced electrical circuit). When the sample passes through the filament there is a different heat conduction through the filament itself. The temperature of the filament varies and therefore also the resistance. At this point, the bridge is electrically unbalanced. The electrical imbalance is amplified and sent to the interface. The signal supplied by the detector is directly proportional to the concentration of the analytes.
The sensitivity of the TCD depends on:
– difference in thermal conductivity between the gas carrier and the analyte;
– analyte concentration;
– filament current.
B) Flame Ionization Detector also called FID. It is a destructive detector. The carrier, conveyed into a nozzle, is mixed with air and hydrogen, which feed a flame placed at the exit of the nozzle. The flame produces a ionic current which is converted into voltage, amplified and processed. When an analyte, such as CH4, reaches the flame is burned and ionized, therefore varying the current intensity and allowing the detection of the signal. The FID is powered by hydrogen produced by a generator and by previously dried air, filtered from hydrocarbons and impurity.

Fig.3
The INGV-Palermo laboratory has the following gas chromatographs:
– 2 Agilent 7890 gas chromatographs equipped with a micro additional gas chromatographic module (Fig.4). The system has been assembled specifically according to the INGV-Palermo requests.
The gaseous sample is introduced within the introduction system, where it is separated into two aliquots.
A first aliquot is introduced into the micro gas chromatographic module, where a PPU capillary column separates the main components. He is used as a carrier gas. A micro TCD detector detects the concentrations of the following gaseous components: CH4, CO2, C2H6, C3H8, H2S, SO2. The second aliquot fills the loop, which is part of the Agilent 7890 gas chromatograph (flushed by carrier Ar) being subsequently introduced in a PPU column. In this column the group of light components (He, H2, O2, N2, CH4, CO) is separated from those characterized by higher elution times (CO2, C2H6, C3H8, H2S, H2O, SO2, etc). The lightweight components group passes through a valve into a Molsieve 5A capillary column, housed in the same compartment of the PPU column (Fig.5). At a preset time, the valve itself cuts the heavier components, that escape in backflush from the PPU, do not reach any detector, and therefore are not quantified. The light components are separated in the MolSieve 5A column, whose exit is connected to a TCD detector system + Methanizer + FID where they are quantified. The TDC detects He, H2, O2, N2, CH4, the FID detects CH4 e CO.
From a single introduction the so-composed system allows the analysis and quantification of the following gaseous species: He, H2, O2, N2, CH4, CO, CO2, C2H6, C3H8, H2S, SO2. The micro gas chromatographic system works in isothermal, while the gas chromatographic one can work in isothermal or in programmed temperature.
– 1 Perkin Elmer Clarus 500 gas chromatograph, Ar-flushed and equipped with a packed column (100/120 Shincarbon, 3 m, Fig. 6 and 7). The Shincarbon columns are composed of carbon spherical microparticles with precise particle size characteristics, constant and reproducible. They are constituted by a carbonaceous skeleton, residue of pyrolysis of a polymeric precursor and they have very high surface area. In addition to the large surface area, other essential features for a good stationary phase, are: a low friability, a high homogeneity of particles dimensions, and a great chemical inertness. The sample is introduced in column using multiport valve and analyzed with the TCD detector system + Methanizer + FID: the analyzed species are He, H2, O2, N2, CO, CH4, CO2.
– 1 Perkin Elmer Autosystem XL gas chromatograph (Fig.8), equipped with a capillary column (RT Msieve 5A, 30 m, 0.53 mm ID) flushed with He carrier. Msieve 5A are columns with molecular sieves consisting of zeolites (aluminum silicates and sodium which may also contain calcium ions). At the column exit a TCD detects the following analytes: Ar, O2, N2, CH4.
-1 Shimadzu 2010 gas chromatograph (Fig.9), with He carrier and equipped with a capillary column CP Poraplot Q and FID detector.
This istrument is used for the quantification of light hydrocarbons (from C1 to C6).



















